Lithium Ion Batteries are a key component in modern technology. They power everything from smartphones to electric vehicles. Understanding how these batteries work is essential for grasping their impact on our daily lives.
These batteries rely on lithium ions moving between the positive and negative electrodes. This movement creates energy, enabling devices to function efficiently. Their compact size and high energy density make them popular in consumer electronics. However, they are not without flaws. Overcharging can cause overheating, and improper disposal poses environmental risks. Such issues demand careful consideration.
Despite their benefits, Lithium Ion Batteries face criticism for their lifecycle. Challenges in recycling and resource extraction raise concerns. As technology evolves, innovations are needed to address these problems. The future of Lithium Ion Batteries depends on balancing efficiency with sustainability. This ongoing dialogue is crucial as we embrace a more energy-dependent world.

Lithium-ion batteries play a crucial role in modern technology. They are widely used in smartphones, laptops, and electric vehicles. According to industry research, the global lithium-ion battery market is expected to reach $100 billion by 2025. This growth reflects the increased demand for energy storage solutions.
These batteries consist of an anode, cathode, and electrolyte. The anode is usually made of graphite, while the cathode contains lithium metal oxides. When the battery charges, lithium ions move from the cathode to the anode through the electrolyte. During discharge, the process reverses, releasing energy. The efficiency of these batteries is substantial, often reaching up to 95%.
However, there are challenges. Lithium-ion batteries degrade over time. This degradation can lead to reduced performance and safety issues. For example, a study found that lithium-ion batteries lose about 20% of their capacity after 500 charge cycles. Companies in the industry are focused on improving longevity and safety. Research continues, aiming for sustainable solutions that can mitigate these challenges.
This chart displays the growth in capacity of lithium-ion batteries over the years. As technology advances, the energy density and efficiency of these batteries have significantly improved, making them crucial for various applications, from smartphones to electric vehicles.
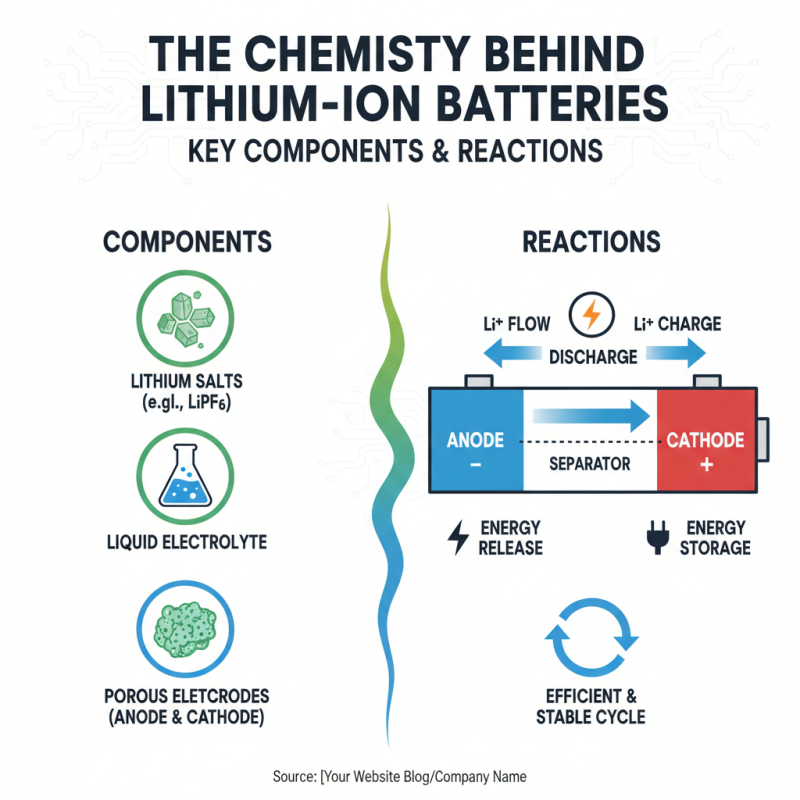
Lithium-ion batteries are an essential part of modern technology. Their chemistry involves several key components that work together to store and release energy efficiently. The primary materials include lithium salts, liquid electrolytes, and porous electrodes. During discharge, lithium ions move from the anode to the cathode, releasing energy. Conversely, during charging, these ions travel back. This flow creates a cycle that is both efficient and relatively stable.
Understanding the reactions in these batteries is crucial. The chemical reactions involve lithium ions intercalating into the anode and cathode materials. Commonly, graphite serves as the anode, while lithium metal oxides are used for the cathode. These materials undergo oxidation and reduction reactions, which are complex yet precise. However, the stability of these reactions can be impacted by various factors, such as temperature and cycle life. Issues may arise, leading to reduced performance or safety risks.
Innovations in battery chemistry are essential for improving performance. Researchers explore new materials to enhance energy density and lifespan. Yet, every solution comes with challenges. The quest for reliable, efficient lithium-ion batteries is ongoing, reflecting the balance between technology and its limitations. This continuous development highlights the importance of understanding the underlying chemistry of these batteries.
Lithium-ion batteries are critical in modern energy storage. They store energy chemically, allowing for efficient charge and discharge cycles. When charging, lithium ions move from the positive electrode to the negative electrode. Upon discharge, they return, releasing energy. This process relies on the movement of lithium ions through an electrolyte, which enables batteries to work effectively.
According to a report by the International Energy Agency (IEA), lithium-ion batteries can achieve over 90% efficiency in energy transfer. This high efficiency stems from their low internal resistance and ability to handle varying power loads. Despite their advantages, these batteries face challenges, such as capacity degradation over time. Research indicates that battery performance can drop by 20% after a few hundred charge cycles.
The size and weight of lithium-ion batteries also play a significant role in their applications. For example, they are now essential in electric vehicles and renewable energy systems. However, the environmental impact of lithium mining and battery disposal raises concerns. Continuous efforts are needed to enhance recycling methods and improve the sustainability of lithium-ion technologies.

Lithium ion batteries have become essential in modern technology. Their performance is primarily assessed through three metrics: capacity, cycle life, and energy density. Capacity measures how much energy a battery can store. It is typically expressed in ampere-hours (Ah) or milliampere-hours (mAh). A higher capacity means longer runtimes for devices. However, this can lead people to assume bigger is better, which isn’t always the case.
Cycle life refers to the number of complete charge and discharge cycles a battery can undergo before its capacity drops significantly. Many users overlook this aspect. A battery with a high capacity but low cycle life may not be practical for daily use. Energy density, on the other hand, measures the amount of energy stored relative to the weight and volume of the battery. Higher energy density allows for smaller and lighter battery designs. However, advancements in energy density may lead to trade-offs in safety and stability. These factors highlight the complexities behind choosing the right lithium ion battery for specific applications. Balancing capacity, cycle life, and energy density requires careful consideration.
Lithium-ion batteries have become essential in today's technology landscape. These batteries power everything from smartphones to electric vehicles. Their ability to store large amounts of energy in a compact size makes them indispensable. In the renewable energy sector, they play a crucial role in managing solar and wind energy. These sources are intermittent, but lithium-ion batteries help store excess energy for later use.
In healthcare, lithium-ion batteries are used in portable devices like insulin pumps and heart monitors. These devices require reliable power sources to ensure patient safety. The efficiency of lithium-ion technology enhances the functionality of the devices, but challenges still remain. Battery lifespan and degradation are concerns that need ongoing attention. Research continues to find solutions to enhance battery performance and safety.
The role of lithium-ion batteries in electric vehicles (EVs) is noteworthy. They enable long-range travel and quick charging. Yet, the production process raises environmental questions. The extraction of lithium has prompted discussions around sustainability and ethical sourcing. Industry leaders are exploring improved recycling methods to address these issues, bringing innovation and responsibility to the forefront of battery technology.






